The Coronavirus has taken the world by storm and there is widespread panic as well as massive health concerns regarding the spreading of this disease. The Coronavirus basically comes from the family of viruses that are known to lead to illnesses like common cold, flu, middle east respiratory syndrome and severe acute respiratory syndrome. One of the new viruses that was recently discovered is the SARS2 Coronavirus (SARS – COV- 2) and the disease caused by this virus is known as COVID -19.

There have been numerous cases of COVID-19 that have been reported from across the world and the number of those diagnosed with this disease is only increasing on a daily basis. One of the reasons for the huge concerns amongst the medical professionals is the fact that the COVID-19 spreads conveniently from people to people.
Education, awareness and early detection are key in effectively fighting the spread of this disease. Hygiene is one of the most important factors that is essential in countering the Coronavirus. Owing to the spread of the disease in multiple countries and continents the various health bodies and organizations such as World health organization (WHO) as well as the US center for disease control and prevention (CDC) are closely monitoring and keeping a watch on the situation and also at the same time are providing rolling updates to keep up with the surmounting challenges posed by the Coronavirus.
Understanding recombinant Coronavirus antigens
The recombinant antigens reagents are also known as COVID-19 protein or Coronavirus protein. The COVID-19 is essentially a novel category of pathogenic Coronavirus, also called SARS CoV2, induced disease which first emerged in China and subsequently has spread rapidly across the globe. The GeneMedi produces types of the recombinant Coronavirus antigens such as Spike protein, Recombinant Nucleocapsid, Spike protein – S1 protein, Envelope E Protein, Spike RBD protein and 3C like Proteinase.
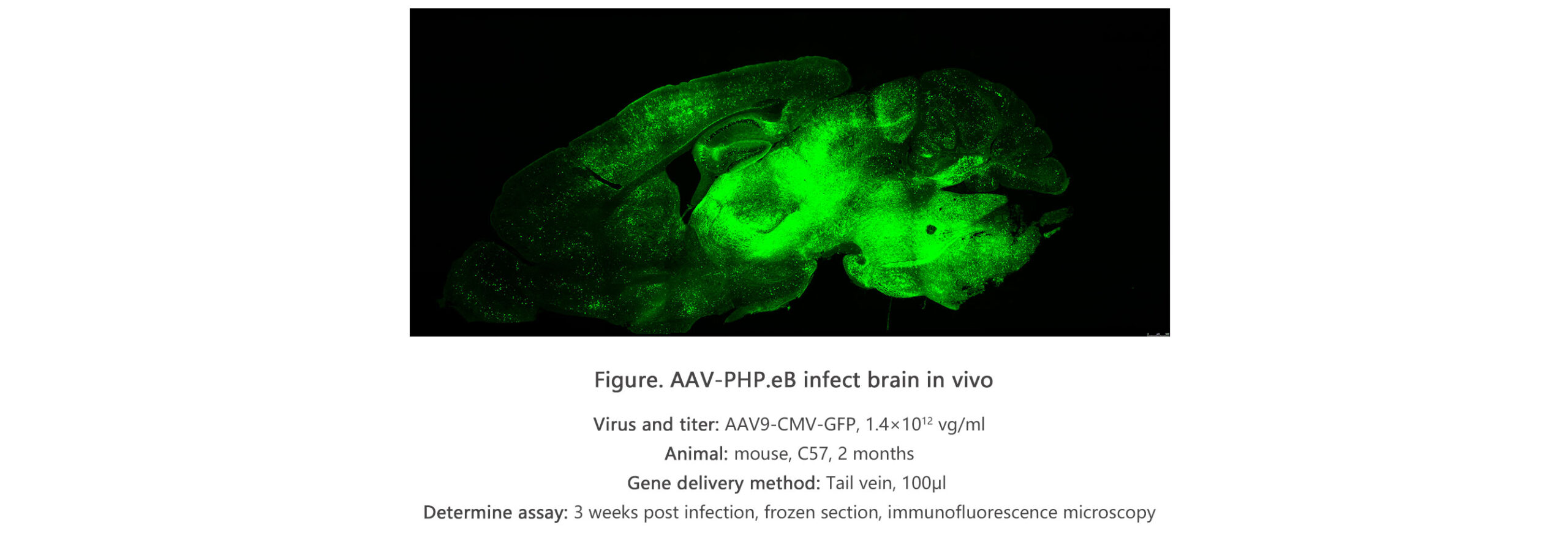
The recombinant proteins related to the Coronavirus were expressed in the types of expression systems which includes mammalian (human cells, HEK293), baculovirus-insect cells and E-Coli. The Coronavirus 2019 recombinant proteins are suitable for the immunogens in the functional ELISA, lateral-flow tests as well as other immunoassays. The GeneMedi has submitted the review of the pre-prints regarding recombinant 2019 nCOV and COVID-19.
The GeneMedi contribution
The GeneMedi was founded in the year 2010 and it is a biotech company that has high expertise in the viral vectors of the transfer in mediated gene, gene therapy and the gene editing. The professionals from GeneMedi provide assistance to the scientists from vector optimization to the academic and also provide help in process development, technology transfer and scalable production.
The scientists at https://www.GeneMedi.net/ offer high quality of scalable production in the adeno-associated virus known as (AAV) vector, adenovirus vector and lentivirus vector. The scientists also provide verified and optimized viral vector mechanisms or systems in the form of lentivirus, AAV and adenovirus. The scientists also provide optimization for novel AAV generation, AAV cassette and capsid evolution. This means creating lesser amount of vectors of immunogenic AAV with the improved expression properties and improved transduction.
The GeneMedi is one of the best in the industry because it has established the required standards of quality control system plus has passed ISO9001:2009 certification of quality technical body. Besides this the company also has copyrights for 15 software, authorization of 1 patent as well as successful application for the invention of 6 patents and the patents for 6 utility models. The company provides extremely professional and technical service for the research in gene function. Besides this GeneMedi also provides high quality and honest services for the pharmaceutical companies and scientists in the department of life sciences. There have been in excess of 200 articles which have used the products and services of GeneMedi.

In terms of the company’s offerings, GeneMedi provides professional service of packaging technology related to adenovirus, lentivirus and adeno-associated virus (AAV). The different recombinant viral systems of packaging are created for the customers. The viruses from the GeneMedi are inspected and tested for ensuring high quality and excellent purity. The viral vectors that are produced by GeneMedi are fit for usage in both the vivo and vitro thereby meeting the maximum demands from the clients and customers. The GeneMedi has also manufactured and launched a large number of the human full length protein coding ORF cDNA clones in the way of various sets for vector systems that come with multiple features that make the clones fit for numerous functional assays, easy purification in cell types, translation coupling systems and cell free transcription and proteomics study and wide scale functional genomics.
If you want to order products or services at GeneMedi then you can do so through phone, email or directly through the website.